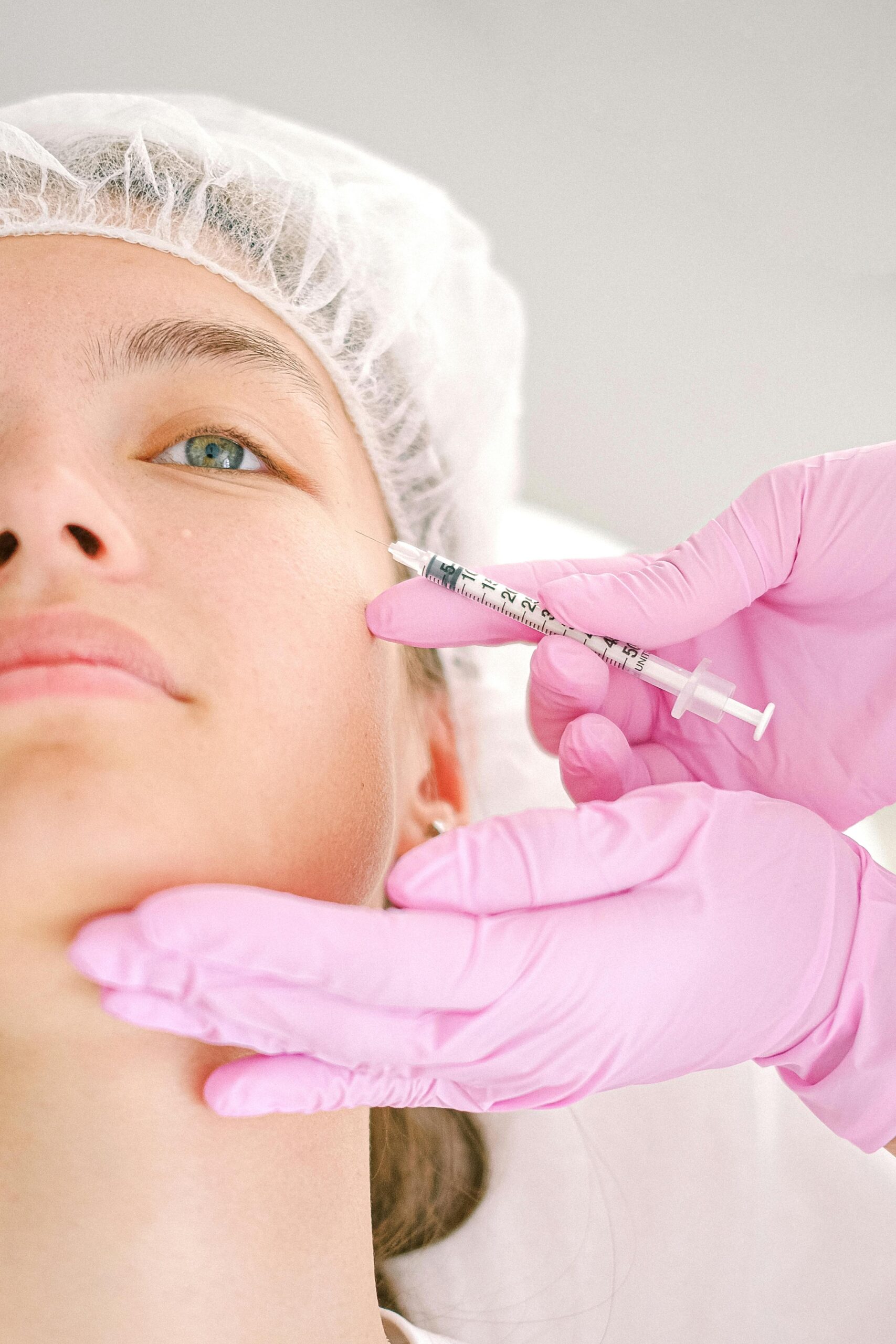
Dermal Fillers
Dermal Fillers
Benefits
Duration
The Process
A simple, step-by-step process designed for your comfort and convenience.

Affordable, Honest Pricing
Pricing varies by product type and treatment area
- VIP pricing available
- Retail pricing available
Final pricing is reviewed during your appointment.
Your Questions, Answered
Common questions to help you feel informed and confident.

